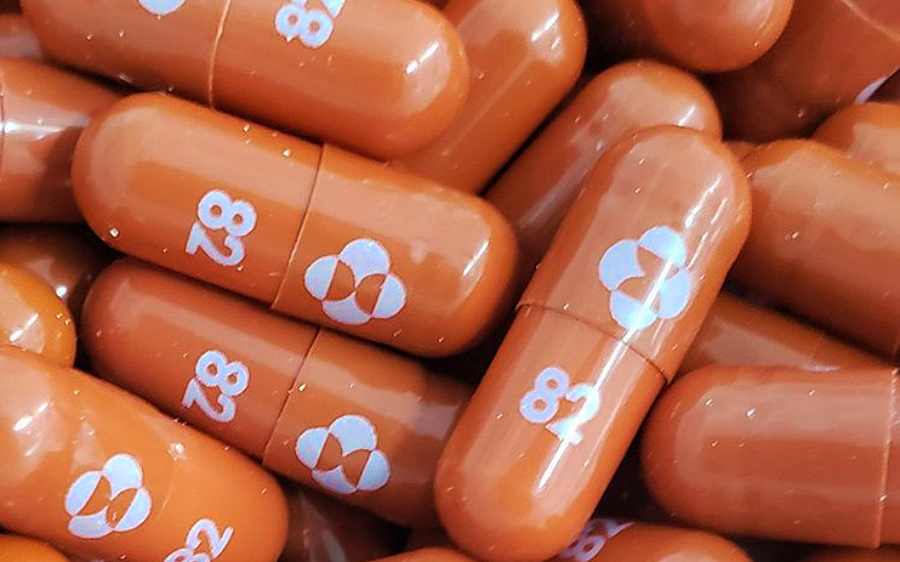
EU REGULATOR STARTS REAL-TIME REVIEW OF MERCK’S COVID-19 PILL
Mon 25 October 2021: The European Union’s medicines authority has started a real-time review of Merck & Co Inc’s experimental COVID-19 antiviral drug for adults, the US firm said on Monday. The European Medicines Agency (EMA) would analyze data as soon as it became available, rather than waiting for a formal application when all essential […]
Continue Reading