Sat 23 December 2023:
OZEMPIC users are being advised by federal officials to verify the authenticity of their drugs due to the sale of counterfeit versions of the well-known diabetic treatment through legitimate channels.
In an ongoing investigation, the Food and Drug Administration has confiscated “thousands of units” of fake Ozempic 1 milligram injections, the agency announced in a consumer notice on Thursday. The notification indicates that there have been five reports of illness connected to the medications, but none of the instances were severe.
Ozempic — which hasn’t even been approved by the FDA as a weight-loss solution — is a once-weekly shot initially designed to treat Type 2 diabetes.
However, it has gained immense mainstream popularity this year for its “miraculous” pound-shedding effects, as shown off by celebrities like Amy Schumer, Chelsea Handler and Tracy Morgan, and endorsed by Oprah, whose Weight Watchers company recently acquired Sequence, a tele-health subscription service that prescribes Ozempic.
Clinicians and patients are advised to check the product packages they have received and not to use those labelled with lot number NAR0074 and serial number 430834149057. Some of these counterfeit products may still be available for purchase, the FDA said in a statement.
Together with Ozempic manufacturer Novo Nordisk, the FDA is investigating “thousands of units” of the 1-mg injection product. Information is not yet available regarding the drugs’ identity, quality, or safety. However, the pen needles have been confirmed as fake — thereby raising the potential risk for infection — as have the pen labels, accompanying healthcare professional and patient label information, and carton.
“FDA takes reports of possible counterfeit products seriously and works closely with other federal agencies and the private sector to help protect the nation’s drug supply. FDA’s investigation is ongoing, and the agency is working with Novo Nordisk to identify, investigate, and remove further suspected counterfeit semaglutide injectable products found in the US,” the statement says.
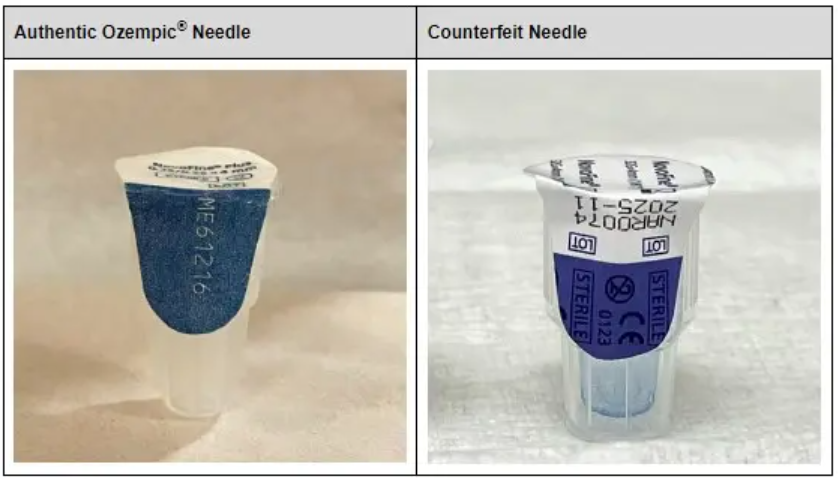
The FDA shared this photo of the counterfeit product, which was initially created to treat Type 2 diabetes but has since gained immense mainstream popularity for its weight-loss effects.
Patients are advised to only obtain Ozempic with a valid prescription through state-licensed pharmacies and to check the product before using for any signs of counterfeiting. There are several differences between the genuine and counterfeit products in the way the pen needle is packaged. The most obvious is that the paper tab covering the fake needle says “Novofine®” whereas the genuine one says “Novofine® Plus.”
There have been at least five adverse events reported from this lot; none were serious and all were consistent with gastrointestinal issues known to occur with the genuine product.
Counterfeit products should be reported to the FDA ‘s consumer complaint coordinator or to the criminal activity division.
SOURCE: INDEPENDENT PRESS AND NEWS AGENCIES
______________________________________________________________
FOLLOW INDEPENDENT PRESS:
WhatsApp CHANNEL
https://whatsapp.com/channel/0029VaAtNxX8fewmiFmN7N22
![]()
TWITTER (CLICK HERE)
https://twitter.com/IpIndependent
FACEBOOK (CLICK HERE)
https://web.facebook.com/ipindependent
Think your friends would be interested? Share this story!